An Autism Drug Got a White House Boost. Prescriptions Soon Followed.
Researchers say a 2025 press briefing was followed by a sharp rise in prescriptions for leucovorin, an inexpensive drug now being studied for autism.
An old drug, a new spotlight, and a sudden shift in autism treatment
For years, Mason Connor’s parents wondered if their son would ever speak. Mason was diagnosed with autism at age 2½ and struggled to communicate despite years of therapy.
Then something unexpected happened.
Three days after Mason started taking a little-known drug called leucovorin, his parents say he said his first word.
I first reported Mason’s story for CBS News in February 2025.
Now, a new study in The Lancet suggests stories like Mason’s may help explain a sudden rise in prescriptions for the drug across the United States.
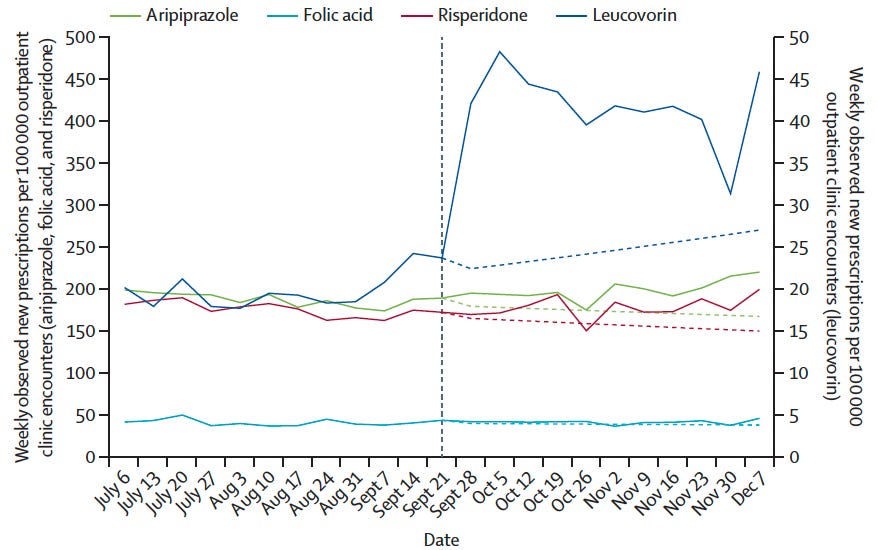
After a White House press briefing in September 2025 highlighted leucovorin as a possible treatment for autism, prescriptions for the drug among children jumped more than 70% above normal levels, according to the study.
The findings show how quickly public attention can influence the way doctors prescribe medicines.
When a presidential briefing moves the medical needle
Changes in medical treatment usually happen slowly. Doctors often wait for large studies and official guidelines before using new treatments.
But the White House briefing appeared to speed things up.
During the event, the president and federal health officials warned about possible links between acetaminophen use during pregnancy and neurodevelopmental disorders. They also spoke about leucovorin as a promising treatment for some children with autism.
Speakers described the drug as “an exciting therapy that may benefit large numbers of children who have suffered from autism.”
Some autism researchers warned that the evidence for leucovorin is still early and that larger studies are needed.
Still, many doctors soon began hearing from parents asking about the drug.
For families searching for ways to help their children communicate, even early signs of a possible treatment can bring hope.
Mining medical records for evidence of change
To understand what happened after the briefing, researchers looked at a large national medical database called Cosmos. The database includes health records from more than 294 million patients treated at hospitals and clinics that use Epic electronic medical records.
Researchers compared prescription trends before and after the White House briefing.
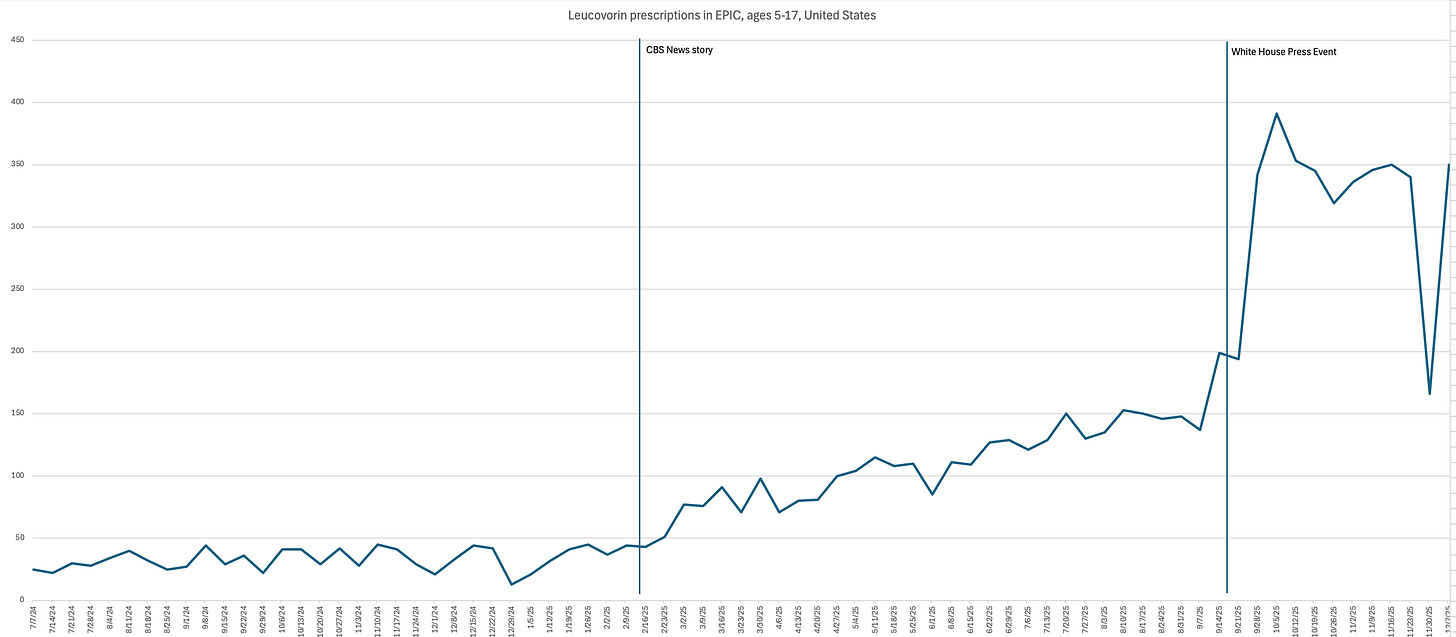
They found that prescriptions for leucovorin in children ages 5 to 17 rose sharply. In the weeks after the briefing, new prescriptions were 71% higher than expected.
The biggest jump happened during the first month after the announcement, when prescriptions nearly doubled.
Out of curiosity, I asked the researchers to see if there was a similar bump in leucovorin prescriptions after my CBS News story originally aired in February 2025. Here’s what they found:
Between February and November 2025, there was an increase, albeit slower, in leucovorin prescriptions.
The White House press briefing also appeared to affect another medication.
Emergency departments gave acetaminophen to pregnant patients about 10% less often than before. This suggests doctors may have been reacting to warnings about possible risks.
Researchers stressed that acetaminophen is still widely considered safe when used as directed, including during pregnancy.
They also found no similar changes in several other medicines, suggesting the shift was tied to the White House briefing.
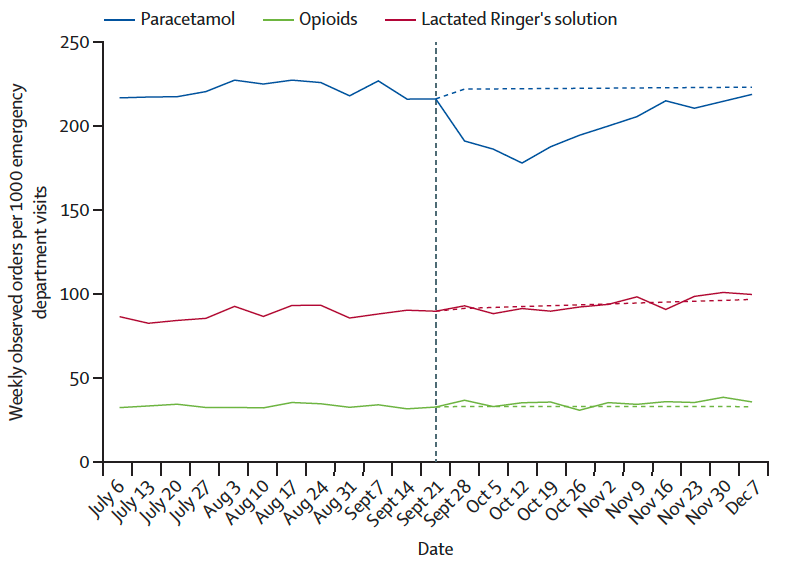
Crucially, the researchers found no similar changes in several comparison drugs, indicating the shifts were likely tied specifically to the briefing and related public messaging.
Why researchers are studying leucovorin for autism
Leucovorin has been used for many years to help reduce side effects from some chemotherapy drugs.
But scientists are now studying it for another reason.
Some children with autism appear to have a problem called cerebral folate deficiency. This means their brains may not get enough folate, a type of vitamin B that helps the brain develop and function.
In some cases, the body makes antibodies that block a protein that normally carries folate into the brain.
Leucovorin can enter the brain through another pathway. Scientists believe this may help restore folate levels in some children.
Several clinical studies suggest the drug may help improve language and communication in certain children with autism, especially those who test positive for these antibodies. Some experts would go so far as to say that these children don’t have autism but rather a newly recognized neurodevelopmental disorder that used to be lumped in with autism.
The studies of leucovorin so far have been small. Researchers say larger trials are needed to know which children might benefit most.
The promise and the limits of early evidence
Mason’s story helps explain why interest in leucovorin has grown so quickly.
Doctors sometimes prescribe drugs “off label,” meaning they use them for conditions that are not officially approved by the Food and Drug Administration. Experts estimate that 20% to 30% of prescriptions fall into this category.
Autism is one area where families and doctors are searching for better treatments.
While some medications can help with symptoms such as irritability, none are approved to treat the core social and communication challenges of autism.
The new Lancet study shows how quickly attention — from the media, government leaders, or advocacy groups — can affect medical decisions.
But experts say new treatments should still be tested carefully in large studies.
For now, leucovorin remains a promising but still developing area of autism research.
And for families like the Connors, the search for answers continues.