Ibogaine: We're Funding Research While the Experiment Runs Unsupervised
The White House wants to study a psychoactive drug that may help veterans with PTSD and addiction. Americans are already taking it at unregulated clinics abroad. At least 27 have died.
More here: https://www.cbsnews.com/news/psychedelic-drug-ibogaine-ptsd-trump-to-sign-executive-order/
The ibogaine executive order and the gap it doesn't close
We are about to spend millions studying ibogaine. At the same time, Americans are flying overseas to take it in clinics with no consistent cardiac screening, no monitoring standards, and no obligation to report outcomes.
In medicine, that sequence is backwards.
The White House is expected to sign an executive order directing federal agencies to fund research into ibogaine, a psychoactive compound derived from a West African shrub. The drug remains Schedule I, grouped with heroin and defined as having no accepted medical use.
The administration says it wants to determine whether ibogaine is legitimate medicine or “snake oil.”
Patients are traveling to clinics in Mexico and the Caribbean, paying thousands of dollars for treatments that can last more than 24 hours and involve intense hallucinations. These clinics operate outside U.S. regulatory oversight. There is no standardized cardiac screening, monitoring varies, and follow-up is inconsistent.
At least 27 deaths have been documented.
As a physician, this is the central concern: not just that ibogaine is being used, but that it is being used in settings that would not meet basic safety standards in the U.S.
The evidence is thin
Ibogaine draws interest because it appears to have real effects, particularly for addiction and, more recently, PTSD and traumatic brain injury.
A 2024 study in Nature Medicine followed 30 Special Operations veterans treated with ibogaine and intravenous magnesium. One month later, symptoms of PTSD, depression, and anxiety had dropped by roughly 80-90%. No serious cardiac events were reported.
But the study had no control group, no placebo, and no blinding. It included a small, highly selected group and followed them for only one month. The authors themselves called for controlled trials to establish safety and efficacy.
That limitation matters more than the results.
Across the literature, rigorous evidence is nearly absent. There is one randomized controlled trial, involving 20 patients with cocaine dependence, published in 2014, with significant methodological gaps. It remains the only controlled trial.
Everything else — observational studies, case reports, surveys — points in the same direction: possible benefit, uncertain magnitude, and real risk.
A 2023 review found some efficacy in reducing withdrawal and cravings. It also identified multiple deaths and described ibogaine’s cardiac toxicity as a serious concern.
The evidence is not conflicting. There just isn’t much of it.
The risk is cardiac
Ibogaine’s primary danger is its effect on the heart.
It disrupts the ion channels that regulate cardiac repolarization, increasing the risk of torsades de pointes, a potentially fatal arrhythmia that can progress to cardiac arrest. This risk is well documented.
Ibogaine is also metabolized into noribogaine, which persists in the body long after the acute effects end, extending the window of risk.
In the 2024 study, magnesium may have stabilized cardiac rhythms. No serious events were observed in that small cohort. Encouraging, but a long way from establishing safety.
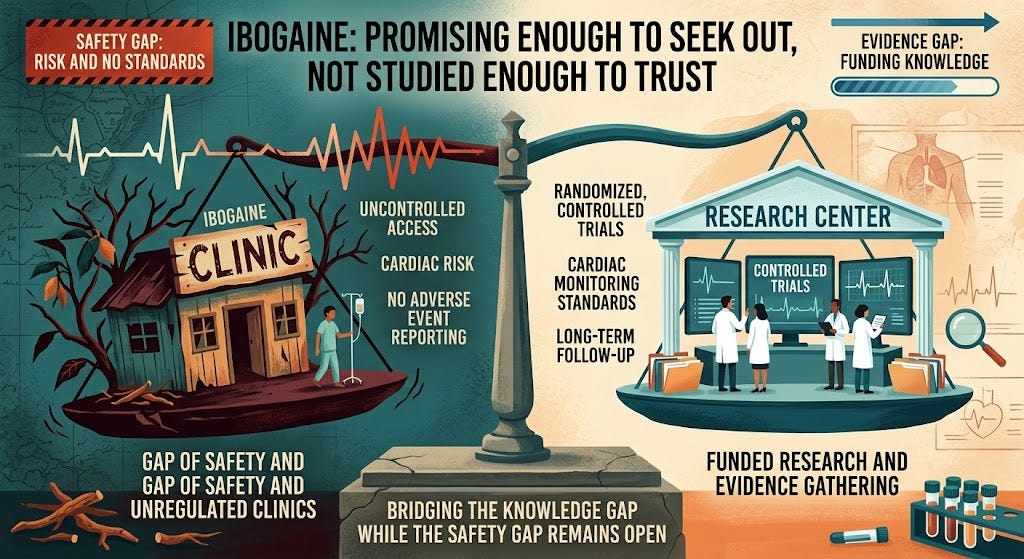
Closing the evidence gap while the safety gap stays open
The executive order focuses on funding research, particularly for veterans. That is necessary. Texas has already committed $50 million to similar efforts.
But this addresses the knowledge gap, not the safety gap.
Ibogaine remains illegal in the U.S. Clinics abroad remain unregulated. There is still no standardized cardiac screening, no consistent monitoring, and no requirement to report adverse events.
So while the government prepares to study ibogaine in controlled settings, patients continue to access it in uncontrolled ones.
What it would take to know whether ibogaine works
Evaluating ibogaine is not conceptually difficult. It requires randomized, placebo-controlled trials with adequate sample sizes and diverse participants. Cardiac monitoring must be built into study design. Follow-up needs to extend beyond weeks.
Two questions need clear answers:
Does ibogaine work better than placebo or existing treatments?
Can its cardiac risks be managed to an acceptable level?
Right now, the answer to both is: we do not know.
Promising enough to seek out, not studied enough to trust
In medicine, the sequence is supposed to be: test first, then treat.
With ibogaine, that sequence has been reversed. Patients — many with severe addiction or PTSD — are pursuing a treatment that is promising enough to seek out but not well enough studied to trust. They are doing so in settings without the safeguards we would require for any drug with known cardiac risk.
An executive order promising future research does not change that. It acknowledges, belatedly, that the science is lagging behind the practice.